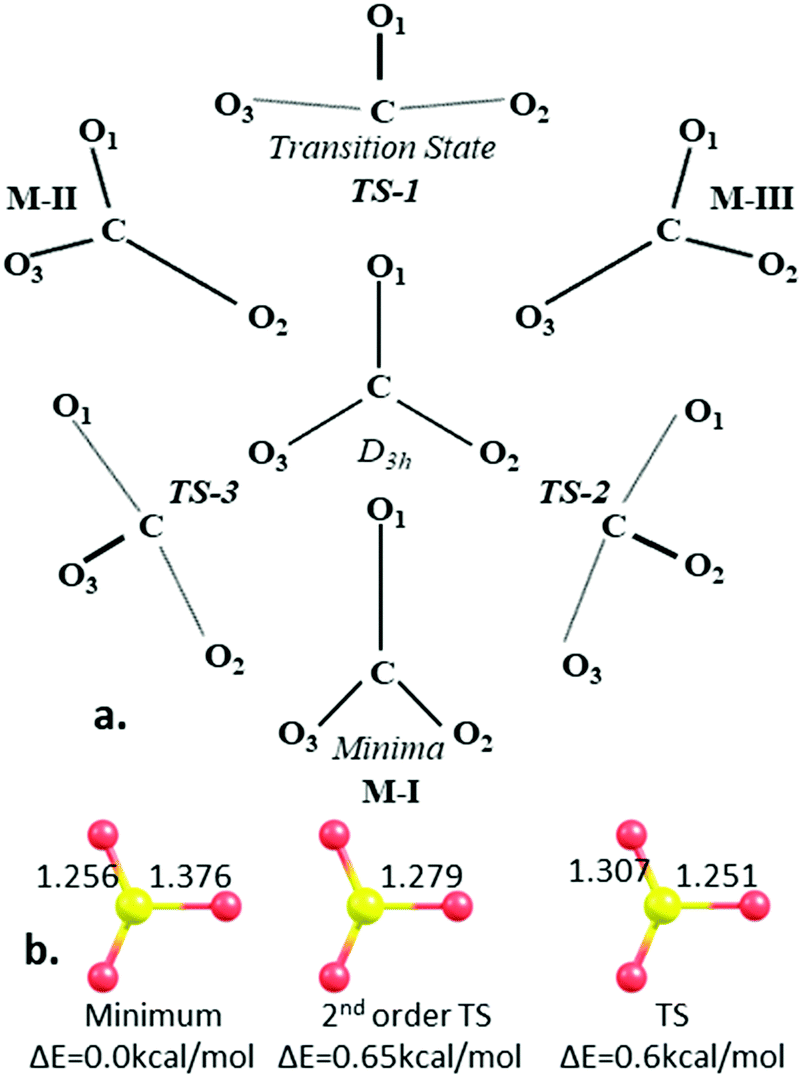
Concentration of the HCO3 − /CO3 2− ions in the formation solution at... | Download Scientific Diagram

Concentration of the HCO3 − /CO3 2− ions in the formation solution at... | Download Scientific Diagram

Chemistry Help: The carbonate ion (CO3 2-) acts as a Bronsted base with water and equations - YouTube

Concentration of the HCO3 − /CO3 2− ions in the formation solution at... | Download Scientific Diagram